Catheter-Directed Thrombolysis vs Low-Dose Alteplase for acute PE
Or are they the same thing?
After the publication of HI-PEITHO, which concluded that catheter-directed thrombolysis for acute PE prevents “cardiorespiratory decompensation or collapse” (a semantically exaggerated result the industry-funded authors cleverly parsed out of the observed improvement in physiology)—
Catheter-Directed Thrombolysis vs. Anticoagulation Alone for PE (HI-PEITHO)
Catheter-directed thrombolysis has been used for more than 30 years as a therapy for acute pulmonary embolism.
—and the endorsement of CDT (but moreso thrombectomy) for PE by multiple professional societies—
Catheter-based interventions for PE are blessed by AHA, ACCP and friends
Through its periodically updated guideline statements, the American College of Chest Physicians has traditionally defined standard care for acute pulmonary embolism. (PulmCCM is not affiliated with any specialty society.)
—intensivists around the world (but especially the U.S., where financial motivations drive a large proportion of care decisions) are asking:
“How can I help my patients with PE avoid undergoing unnecessary procedures?”
Most critical care folks know of Dr Josh Farkas and his famed PulmCrit blog and online textbook. If not, you should check them out.
Farkas recently gave an enlightening take on recent trials and CDT generally, arguing that catheter-directed thrombolysis is (in all important respects) really just peripherally-delivered low-dose tPA with a useless and expensive plastic apparatus attached, whose purpose is to treat the prescribing clinician’s anxiety about causing bleeding with “systemic tPA” (which, due to the circulatory nature of the circulatory system, catheter-directed tPA is, too).
Others made comments about the trial in the same spirit: I wish there had been a low-dose tPA arm in HI-PEITHO.
As it turns out, the recently published STRATIFY trial included just such a comparison.
Instead of reinventing wheels, let’s take Farkas’s arguments in these great posts on HI-PEITHO (here) and STRATIFY (here) out for a spin.
What Are Low-Dose Thrombolytics for PE?
“Full-dose” thrombolytics include alteplase (tPA), which is typically given as 100 mg IV for massive, life-threatening pulmonary PE, in a 10 mg bolus followed by a 90 mg infusion over two hours. Tenecteplase (TNK) is not FDA-approved for PE, but its full dose is often given as a 50 mg bolus. TNK dosing can also be weight-based up to 50 mg (with ranges of ~30 to 50 mg in the PEITHO trial, e.g.)
“Low-dose” tPA has no standard definition, but 10 mg IV bolus followed by 40 mg IV infused over 2 hours was used in one widely mentioned RCT.
Yet nothing bars careful and thoughtful physicians from using lower doses of tPA (e.g., 25 mg), or slower infusions of any dose. Anecdotally, a minority do.
Reduced-dose TNK has not been well-described in the published literature.
Evidence for Lower-Dose tPA for Acute PE
As with most other treatments for acute pulmonary embolism, there is very little evidence from randomized trials to support the use of reduced-dose tPA.
The MOPETT trial (Am J Cardiol 2013) enrolled 121 patients at one center in Arizona, randomizing them to 50 mg tPA with anticoagulation or AC alone. Although all patients were symptomatic and had elevated estimated pulmonary artery pressures on echocardiogram, the authors did not report how ill the patients were at enrollment (not even their BP), nor after treatment. Low-dose tPA resulted in lower estimated PA pressures and recurrent PE. Four patients died (one in the tPA, three in the AC only arm). Authors reported zero bleeding in either arm (this is unusual). They dubbed their intervention “safe dose tPA” in their methods (before getting results). Investigators were unblinded. The risk of bias is high. MOPETT is minimally informative by today’s standards, and hard to compare to other trials in PE.
A RCT by Wang et al. (n=118), performed in China and published in Chest in 2010, concluded that 50 mg tPA produced outcomes equivalent to 100 mg tPA, with less bleeding.
The STRATIFY trial (n=210) (Cardiovascular Research 2026) suggested 20 mg of peripherally infused tPA over 6 hours worked better than heparin and as well as CDT at reducing radiographic clot burden. Bleeding was numerically higher (equally) with either low-dose lytics or CDT as compared to heparin alone. Mortality was low, few PEs became severe, and the trial was not powered to detect differences in clinical outcomes.
PEITHO-3 is testing low-dose tPA (0.6 mg/kg IV up to 50 mg over 15 minutes) against placebo (plus heparin in both arms) in intermediate-high risk PE. It doesn’t include a CDT arm. Results are expected around 2027.
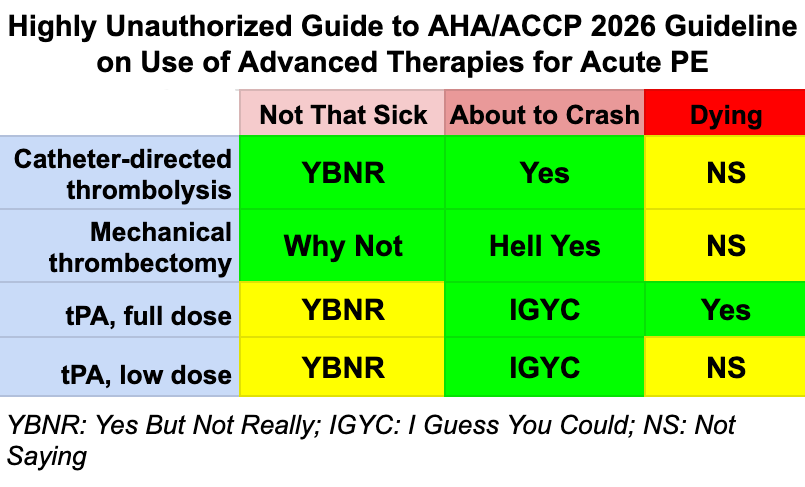
Guidelines Endorse Low-Dose tPA, Anyway
As we pointed out recently, guidelines co-signed by multiple societies (AHA, ACC, ACCP, ACEP, etc.) endorsed virtually all non-insane approaches to treating acute pulmonary embolism, regardless of the evidence base (which was low for all therapies).
Low-dose tPA got its seat at the table with a 2b (weak) recommendation for many patients with intermediate-high risk PE.
PulmCCM is not affiliated with any specialty society.
Which brings us to Farkas’s most interesting argument.
The Claim: Catheter-Directed tPA ≈ Very Low-Dose Systemic tPA
First, let’s compare apples to apples:
During catheter-directed thrombolysis using Boston Scientific’s ultrasonic-tipped EKOS™ catheter in HI-PEITHO (n=544), about 8 mg tPA was instilled into each pulmonary artery (usually both, ~16 mg total) over about 7 hours. The control arm got heparin, not peripheral tPA.
In STRATIFY, 20 mg tPA was administered over 6 hours, systemically or using the EKOS catheter (or neither in the heparin-only arm). Clot dissolution was statistically equivalent between the CDT and systemic low-dose tPA arms (with methodologic problems noted here). With fewer than 80 patients per arm who were not that sick, the study lacked statistical power to identify clinically meaningful differences.
Bleeding rates were numerically and statistically similar between peripherally-infused and centrally-infused tPA.
Sub-Claim #1: Instillation of tPA in the pulmonary artery does not lyse clot any more effectively than peripheral tPA.
This is supported by a study by Verstraete et al in 1988, showing equivalent results after either form of tPA administration, although in only 34 patients.
This was followed by an in vitro experimental study in dogs by Schmitz-Rode (Cardiovasc Intervent Radiol 1998), which showed what you would intuitively predict: intrapulmonary artery-instilled liquid simply flowed down the path of least resistance, away from the PA clots, into the systemic circulation.
Subsequently, Boston Scientific added an ultrasound tip to its catheter that purports to force tPA sideways into the clot.
Sub-Claim #2: Ultrasound “assistance” doesn’t increase penetration into the clot or provide any advantage over conventional catheters (which aren’t better than peripheral tPA, either)
From PulmCrit:
Over time, it became increasingly clear that ultrasonic energy is ineffective. Some studies in DVT showed this, as well as a registry trial in pulmonary embolism. (25856269, 27630267, 28827014, 25593121, 26993702, 30915914, 30915912) More recently, the SUNSET trial compared USAT to conventional catheter-directed thrombolysis and found no differences. (34167677)
Thus, the entire theoretical rationale for using catheter-directed thrombolysis collapsed. If Verstraete et al. showed that catheter-directed thrombolysis was worthless, and ultrasonic energy is worthless then… the entire intervention makes no sense.
The Verdict:
It is as likely as not that peripherally-infused tPA is equally effective to catheter-directed tPA for acute PE, at equal doses and infusion rates. I.E. there is equipoise on this question.
This is in no small part because device manufacturers have (shrewdly) shied away from testing their catheters directly against peripherally infused tPA.
With STRATIFY providing the only recent and relevant data, until larger trials testing CDT against peripheral low-dose tPA are performed, there is no good evidence that CDT is superior to peripheral tPA at equivalent doses.
The Implication:
It seems medically, ethically, and professionally appropriate for clinicians to advocate against catheter-directed thrombolysis in favor of peripherally-infused low-dose tPA at similar doses and infusion duration, because it spares patients with intermediate-high risk PE a potentially unhelpful procedure, while providing likely comparable efficacy at a similar (or lower) risk.
This approach would bring up yet another wrinkle, though.
Assume low-dose tPA (e.g. 18 mg over 7 hours) carries a higher rate of bleeding than thrombectomy (which seems likely), but with the advantage of being noninvasive.
If that is the case, and thrombectomy is at least as effective as low-dose tPA (as also seems likely, in patients with accessible clot)—at what rate of tPA-induced bleeding does a “thrombectomy first” approach—in which many people with intermediate-high risk PE would be unnecessarily catheterized to improve a few and avoid bleeding in a few more—make sense?
This line of thought tends to align with the recent guidelines’ subtle support of thrombectomy over CDT.
Another possible approach: low-dose tPA for most patients with intermediate-high risk PE with abnormal physiology; mechanical thrombectomy as first-line for the more severe cases or as rescue for those who deteriorate. Their bleeding risk for the procedure after tPA might not be markedly higher than if they had gotten catheterized for CDT in the first place.
A significant additional risk from this option? Hearing “I told you so” from the proceduralist who wanted to do it earlier at 2 pm, not now at 3 am.
PEERLESS II (an industry-funded thrombectomy trial) is expected to wrap up in mid-2026.
References
Kjaergaard J, Bang LE, Sonne-Holm E, et al. Randomized trial of low-dose, ultrasound-assisted thrombolysis or heparin for pulmonary embolism. Cardiovascular Research. Published online January 30, 2026. doi:https://doi.org/10.1093/cvr/cvag038
Rosenfield K, Klok FA, Piazza G, et al. Ultrasound-Facilitated, Catheter-Directed Fibrinolysis for Acute Pulmonary Embolism. New England Journal of Medicine. Published online March 28, 2026. doi:https://doi.org/10.1056/nejmoa2516567
Sharifi M, Bay C, Skrocki L, Rahimi F, Mehdipour M. Moderate Pulmonary Embolism Treated With Thrombolysis (from the “MOPETT” Trial). The American Journal of Cardiology. 2013;111(2):273-277. doi:https://doi.org/10.1016/j.amjcard.2012.09.027
Schmitz-Rode T, Kilbinger M, Günther RW. Simulated flow pattern in massive pulmonary embolism: significance for selective intrapulmonary thrombolysis. Cardiovascular and interventional radiology. 1998;21(3):199-204. doi:https://doi.org/10.1007/s002709900244
Sanchez O, Anaïs Charles-Nelson, Ageno W, et al. Reduced-Dose Intravenous Thrombolysis for Acute Intermediate–High-risk Pulmonary Embolism: Rationale and Design of the Pulmonary Embolism International THrOmbolysis (PEITHO)-3 trial. Thrombosis and Haemostasis. 2021;122(05):857-866. doi:https://doi.org/10.1055/a-1653-4699
And of course PulmCrit




I was recently look8ng over the new guidelines and have to chuckle at the criticism of the evidence quality. It would seem that in some cases the guidelines claim a higher strength of evidence than the average physician is appraising the evidence as.
During Covid when suspected PE on some newly hypoxic pts on vent whose peak and plateaus hadn’t changed suspected PE and even gave one near crashing pt TPA 25mg empirically and the hypoxia resolved so quickly - even got extubated. That was due to resource overload (not getting the cta but still).
Like you said a small minority really is finding success via low dose peripheral tpa - especially versus ekos not sure the ekos has much advantage over tpa.
Glad we might have some data.
The most disappointing thing is what outcomes do we care about? You mentioned as such in the last post the dubious utility of the composite 7d outcomes in HIPEITHO - and the harm signal being real. But what’s a reasonable outcome for the trials we’d desire? 90d pulm htn/cteph or mortality? Or acute clinical things?
We are likely never going to know.
My anecdotal last 2-PE both got theombectomy - neither of them needed it but IR insisted in the “pert team where they seem to be head coach”. One patient had a big catheter hematoma and heparin needed to be held and hemoglobin did drop. The other patient did ok - but he was on room air, breathing at 12 breaths/min, bnp normal, normal bp, Trop downtrended after baby bump but rv appeared large on CT. Pt did ok!
Being this annoying “I’m not sure what we are really giving the pt for this procedure” will not play too well in American medicine though.
It’s likely the future. My only issue is that community hospitals that lack active IR will defer perfectly safe and tolerated low dose tpa waiting for transfer to a center that does.
And for sure that Fri 930pm pt - does IR come in for that?